Research Laboratory

Leadership
Jerome Siegel, PhD
Director
Contact Info
https://teams.semel.ucla.edu/sleep-research
Neurobiology Research 151A3
VAGLAHS, North Hills, CA
jsiegel@ucla.edu
1-818-891-8612
About
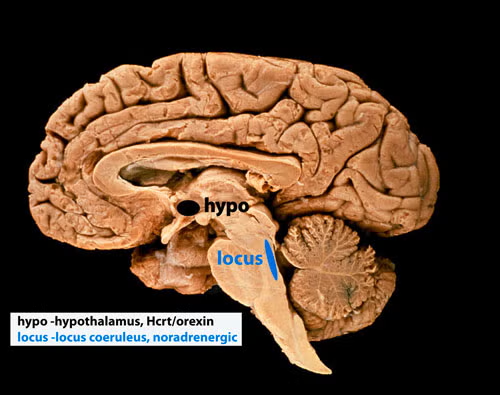
I have been focused on determining the function of sleep and more recently the function of the peptide hypocretin (Hcrt or orexin). We were the first to report the loss of hypothalamic hypocretin neurons in human narcolepsy (March 5, 2000, Society for Neuroscience), the first to record hypocretin neurons in behaving animals (PMID:15924864), finding that these neurons fired in relation to approach/pleasurable behavior (see video link here) and the first to record Hcrt release in the human brain (PMID:23462990), finding greatly elevated Hcrt release during pleasurable social interactions and minimal release during aversion or disappointment. In 2025 we discovered that all humans with narcolepsy also have a loss of locus coeruleus noradrenergic neurons comparable in size to the loss of their hypocretin neurons. Locus coeruleus neurons facilitate muscle tone, thus their loss may underlie cataplexy (PMID:10391445, PMID:11549748, PMID:20668280).
Our studies in 2025 (doi: https://doi.org/10.1101/2025.04.12.648456) show that this loss of locus coeruleus neurons is not caused by the loss of hypocretin neurons. The two neuronal losses, of hypocretin in the forebrain and of norepinephrine in the brainstem locus coeruleus (see below), underlie human narcolepsy and may both be caused simultaneously by an autoimmune process. About 150,000 Americans have narcolepsy, a lifelong disorder.
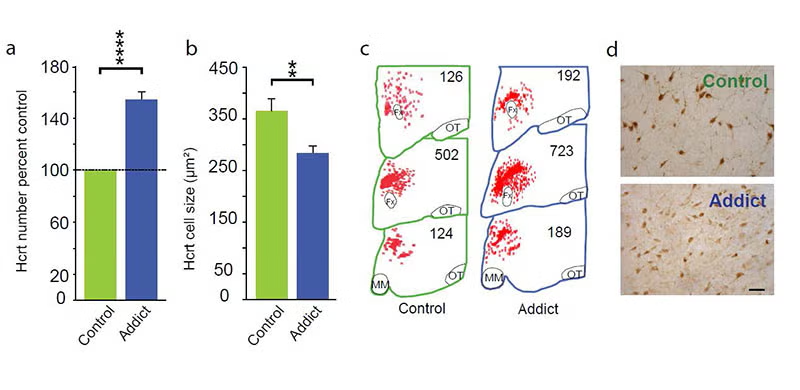
In 2018 and 2024 and 2025 [pmid: 29950444, 39989723, and doi: https://doi.org/10.1101/2025.03.20.644444] we discovered that hypocretin neurons have a major role in opioid addiction. Chronic use of heroin and daily injection of morphine in mice increases the number of detected hypocretin neurons and decreases their size (see figure below) Opioid dependence can be prevented in animals by deleting hypocretin neurons. In 2024 we found that opioid dependence can be prevented, without reducing opioid analgesia, by blocking hypocretin receptors with suvorexant when administering opioids (PMID:39989723). There are two types of Hcrt neuron, only one of which is related to opioid addiction (doi: https://doi.org/10.1101/2025.03.20.644444). About 8 million Americans have opioid dependence. Opioid dependence caused more than 80,000 opioid overdose deaths in the US last year. Most individuals who die from an opioid overdose, had their first exposure to opioids prescribed for analgesia.
Bibliography: https://www.ncbi.nlm.nih.gov/myncbi/jerome.siegel.1/bibliography/public/